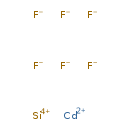Cadmium fluosilicate (CHEM000319)
| Record Information | |||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Creation Date | 2009-03-06 18:58:39 UTC | ||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 2016-11-09 01:08:13 UTC | ||||||||||||||||||||||||||||||||||||||||||||||||
| Accession Number | CHEM000319 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | |||||||||||||||||||||||||||||||||||||||||||||||||
| Common Name | Cadmium fluosilicate | ||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Small Molecule | ||||||||||||||||||||||||||||||||||||||||||||||||
| Description | Cadmium fluosilicate is a chemical compound of cadmium. Cadmium is a transition metal and chemical element with the symbol Cd and atomic number 48. It is found naturally in the earth's crust, though rarely on it's own. (4) | ||||||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Sources |
| ||||||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Type |
| ||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Structure | |||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms |
| ||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula | CdF6Si | ||||||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Mass | 254.487 g/mol | ||||||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Mass | 255.871 g/mol | ||||||||||||||||||||||||||||||||||||||||||||||||
| CAS Registry Number | 17010-21-8 | ||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name | silicon(4+) ion cadmium(2+) ion hexafluoride | ||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional Name | silicon(4+) ion cadmium(2+) ion hexafluoride | ||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES | [F-].[F-].[F-].[F-].[F-].[F-].[Si+4].[Cd++] | ||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Identifier | InChI=1S/Cd.6FH.Si/h;6*1H;/q+2;;;;;;;+4/p-6 | ||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key | IQBPNJJDIXBFNB-UHFFFAOYSA-H | ||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | |||||||||||||||||||||||||||||||||||||||||||||||||
| Description | belongs to the class of inorganic compounds known as transition metal fluorides. These are inorganic compounds in which the largest halogen atom is fluorine, and the heaviest metal atom is a transition metal. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Inorganic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Mixed metal/non-metal compounds | ||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Transition metal salts | ||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Transition metal fluorides | ||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Transition metal fluorides | ||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents | |||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| ||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | |||||||||||||||||||||||||||||||||||||||||||||||||
| Status | Detected and Not Quantified | ||||||||||||||||||||||||||||||||||||||||||||||||
| Origin | Exogenous | ||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations |
| ||||||||||||||||||||||||||||||||||||||||||||||||
| Biofluid Locations | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| Tissue Locations | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| Pathways | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| Applications | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Roles | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Roles | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | |||||||||||||||||||||||||||||||||||||||||||||||||
| State | Solid | ||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | No data. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | |||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra |
| ||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Profile | |||||||||||||||||||||||||||||||||||||||||||||||||
| Route of Exposure | Oral (4) ; inhalation (4) ; dermal (4) | ||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism of Toxicity | Cadmium initially binds to metallothionein and is transported to the kidney. Toxic effects are observed once the concentration of cadmium exceeds that of available metallothionein, and it has also been shown that the cadmium-metallothionein complex may be damaging. Accumulation of cadmium in the kidney results in increased excretion of vital low and high weight molecular proteins. Cadmium is a high affinity zinc analog and can interfere in its biological processes. It also binds to and activates the estrogen receptor, likely stimulating the growth of certain types of cancer cells and causing other estrogenic effects, such as reproductive dysfunction. Cadmium causes cell apoptosis by activating mitogen-activated protein kinases. (5, 1, 2, 3) | ||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolism | Cadmium is absorbed from oral, inhalation, and dermal routes. Cadmium initially binds to metallothionein and albumin and is transported mainly to the kidney and liver. Toxic effects are observed once the concentration of cadmium exceeds that of available metallothionein, and it has also been shown that the cadmium-metallothionein complex may be damaging. Cadmium is not known to undergo any direct metabolic conversion and is excreted unchanged, mainly in the urine. (4) | ||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Values | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| Lethal Dose | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| Carcinogenicity (IARC Classification) | 1, carcinogenic to humans. (8) | ||||||||||||||||||||||||||||||||||||||||||||||||
| Uses/Sources | No data. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Minimum Risk Level | Acute Inhalation: 0.00003 mg/m3 (7) Chronic Inhalation: 0.00001 mg/m3 (7) Intermediate Oral: 0.0005 mg/kg/day (7) Chronic Oral: 0.0001 mg/kg/day (7) | ||||||||||||||||||||||||||||||||||||||||||||||||
| Health Effects | Chronic exposure to cadmium fumes can cause chemical pneumonitis, pulmonary edema, and lung diseases such as bronchitis and emphysema. Cadmium also accumulates in the kidneys, causing permanent damage. Loss of bone density also occurs. (4) | ||||||||||||||||||||||||||||||||||||||||||||||||
| Symptoms | Acute inhalation of cadmium fumes results in metal fume fever, which is characterized by chills, fever, headache, weakness, dryness of the nose and throat, chest pain, and coughing. Ingestion of cadmium causes vomiting and diarrhea. (4) | ||||||||||||||||||||||||||||||||||||||||||||||||
| Treatment | Cadmium poisoning is treated by removal from exposure and supportive care. If ingested, induced vomiting or gastric lavage may be performed. (9) | ||||||||||||||||||||||||||||||||||||||||||||||||
| Concentrations | |||||||||||||||||||||||||||||||||||||||||||||||||
| Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||
| External Links | |||||||||||||||||||||||||||||||||||||||||||||||||
| DrugBank ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| HMDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| FooDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| Phenol Explorer ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| KNApSAcK ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| BiGG ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| BioCyc ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| METLIN ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| PDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| Wikipedia Link | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| Chemspider ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| ChEBI ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| PubChem Compound ID | 197153 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Kegg Compound ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| YMDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| ECMDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| References | |||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| MSDS | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| General References | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||