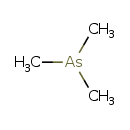Trimethylarsine (CHEM000256)
| Record Information | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Creation Date | 2009-03-06 18:58:29 UTC | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 2026-04-05 17:48:23 UTC | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Accession Number | CHEM000256 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Common Name | Trimethylarsine | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Small Molecule | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | Trimethylarsine is an organic derivative of arsine, as well as the byproduct of microbial action on naturally occuring inorganic forms of arsenic. It is often used in the production of other organoarsenic compounds. Arsenic is a chemical element that has the symbol As and atomic number 33. It is a poisonous metalloid that has many allotropic forms: yellow (molecular non-metallic) and several black and grey forms (metalloids) are a few that are seen. Three metalloidal forms of arsenic with different crystal structures are found free in nature (the minerals arsenopyrite and the much rarer arsenolamprite and pararsenolamprite), but it is more commonly found as a compound with other elements. (10, 7) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Sources |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Type |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Structure | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula | C3H9As | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Mass | 120.025 g/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Mass | 119.992 g/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS Registry Number | 593-88-4 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name | trimethylarsane | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional Name | trimethylarsine | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES | C[As](C)C | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Identifier | InChI=1S/C3H9As/c1-4(2)3/h1-3H3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key | HTDIUWINAKAPER-UHFFFAOYSA-N | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | belongs to the class of organic compounds known as trialkylarsanes. These are organoarsenic compounds containing a trivalent arsenic atom linked to three alkyl groups. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Organic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Organometallic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Organometalloid compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Organoarsenic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Trialkylarsanes | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Aliphatic acyclic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Status | Detected and Not Quantified | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Origin | Exogenous | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biofluid Locations | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tissue Locations | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathways | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Applications | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Roles | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Roles | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| State | Liquid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | Colorless liquid. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Profile | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Route of Exposure | Oral (2) ; inhalation (2); dermal (2) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism of Toxicity | Arsenic and its metabolites disrupt ATP production through several mechanisms. At the level of the citric acid cycle, arsenic inhibits pyruvate dehydrogenase and by competing with phosphate it uncouples oxidative phosphorylation, thus inhibiting energy-linked reduction of NAD+, mitochondrial respiration, and ATP synthesis. Hydrogen peroxide production is also increased, which might form reactive oxygen species and oxidative stress. Arsenic's carginogenicity is influenced by the arsenical binding of tubulin, which results in aneuploidy, polyploidy and mitotic arrests. The binding of other arsenic protein targets may also cause altered DNA repair enzyme activity, altered DNA methylation patterns and cell proliferation. (9, 1) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolism | Arsenic is absorbed mainly by inhalation or ingestion, as to a lesser extent, dermal exposure. It is then distributed throughout the body, where it is reduced into arsenite if necessary, then methylated into monomethylarsenic (MMA) and dimethylarsenic acid (DMA) by arsenite methyltransferase. Arsenic and its metabolites are primarily excreted in the urine. Arsenic is known to induce the metal-binding protein metallothionein, which decreases the toxic effects of arsenic and other metals by binding them and making them biologically inactive, as well as acting as an antioxidant. (3) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Values | LD50: 7870 mg/kg (Oral, Rouse) LC50: 20 500 ppm over 4 hours (Inhalation, Mouse) (8) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lethal Dose | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Carcinogenicity (IARC Classification) | 3, not classifiable as to its carcinogenicity to humans. (6) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Uses/Sources | Trimethylarsine is used in the microelectronics industry, the production of other organoarsenic compounds, and as a ligand in coordination chemistry. (7) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Minimum Risk Level | Acute Oral: 0.005 mg/kg/day (5) Chronic Oral: 0.0003 mg/kg/day (5) Chronic Inhalation: 0.01 mg/m3 (5) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Health Effects | Arsenic poisoning can lead to death from multi-system organ failure, probably from necrotic cell death, not apoptosis. Arsenic is also a known carcinogen, esepcially in skin, liver, bladder and lung cancers. (9, 3) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symptoms | Exposure to lower levels of arsenic can cause nausea and vomiting, decreased production of red and white blood cells, abnormal heart rhythm, damage to blood vessels, and a sensation of burn (9). | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Treatment | Arsenic poisoning can be treated by chelation therapy, using chelating agents such as dimercaprol, EDTA or DMSA. Charcoal tablets may also be used for less severe cases. In addition, maintaining a diet high in sulfur helps eliminate arsenic from the body. (3) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Concentrations | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Links | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| DrugBank ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HMDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FooDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phenol Explorer ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| KNApSAcK ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BiGG ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BioCyc ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| METLIN ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikipedia Link | Trimethylarsine | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemspider ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ChEBI ID | 27130 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PubChem Compound ID | 68978 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kegg Compound ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| YMDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ECMDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MSDS | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| General References | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||