Arsenate (CHEM000195)
| Record Information | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Creation Date | 2009-03-06 18:58:19 UTC | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 2016-11-09 01:08:11 UTC | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Accession Number | CHEM000195 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Common Name | Arsenate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Small Molecule | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | The arsenate ion is AsO43−. An arsenate (compound) is any compound that contains this ion.The arsenic atom in arsenate has a valency of 5 and is also known as pentavalent arsenic or As[V].Arsenate resembles phosphate in many respects, since arsenic and phosphorus occur in the same group (column) of the periodic table. In acidic conditions arsenate exists as arsenic acid, H3AsO4;in weakly acid conditions it exists as dihydrogen arsenate ion, H2AsO4−; in weakly basic conditions it exists as hydrogen arsenate ion HAsO42−; and finally, in basic conditions, it exists as the arsenate ion AsO43−. Arsenate can replace inorganic phosphate in the step of glycolysis that produces 1,3-bisphosphoglycerate, yielding 1-arseno-3-phosphoglycerate instead. This molecule is unstable and quickly hydrolyzes, forming the next intermediate in the pathway, 3-phosphoglycerate. Therefore glycolysis proceeds, but the ATP molecule that would be generated from 1,3-bisphosphoglycerate is lost - arsenate is an uncoupler of glycolysis, explaining its toxicity.[1]. Some species of bacteria obtain their energy by oxidizing various fuels while reducing arsenates to form arsenites. The enzymes involved are known as arsenate reductases. In 2008, bacteria were discovered that employ a version of photosynthesis with arsenites as electron donors, producing arsenates (just like ordinary photosynthesis uses water as electron donor, producing molecular oxygen). The researchers conjectured that historically these photosynthesizing organisms produced the arsenates that allowed the arsenate-reducing bacteria to thrive. (Wikipedia). Arsenic acid is the chemical compound with the formula H3AsO4. More descriptively written as AsO(OH)3, this colorless acid is the arsenic analogue of phosphoric acid. Arsenate and phosphate salts behave very similarly. Arsenic acid as such has not been isolated, but only found in solution where it is largely ionized. Its hemihydrate form (H3AsO4_а__H2O) does form stable crystals. Crystalline samples dehydrate with condensation at 100 C. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Sources |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Type |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
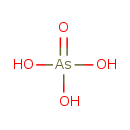
| Chemical Structure | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula | AsH3O4 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Mass | 141.943 g/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Mass | 141.925 g/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS Registry Number | 15584-04-00 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name | arsoric acid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional Name | arsenic acid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES | O[As](O)(O)=O | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Identifier | InChI=1S/AsH3O4/c2-1(3,4)5/h(H3,2,3,4,5) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key | DJHGAFSJWGLOIV-UHFFFAOYSA-N | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | belongs to the class of inorganic compounds known as miscellaneous arsenates. These are inorganic compounds in which the largest metallic oxoanion is arsenate, to which either no atom or a non metal atom is bonded. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Inorganic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Mixed metal/non-metal compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Miscellaneous mixed metal/non-metals | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Miscellaneous metallic oxoanionic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Miscellaneous arsenates | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Status | Detected and Not Quantified | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Origin | Exogenous | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biofluid Locations | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tissue Locations | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathways |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Applications | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Roles | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Roles | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| State | Solid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | White crystals. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Profile | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Route of Exposure | Oral (4) ; inhalation (4) ; dermal (4) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism of Toxicity | Arsenic and its metabolites disrupt ATP production through several mechanisms. At the level of the citric acid cycle, arsenic inhibits pyruvate dehydrogenase. By competing with phosphate it uncouples oxidative phosphorylation, thus inhibiting energy-linked reduction of NAD+, mitochondrial respiration, and ATP synthesis. Hydrogen peroxide production is also increased, which might form reactive oxygen species and oxidative stress. Arsenic's carginogenicity is influenced by the arsenical binding of tubulin, which results in aneuploidy, polyploidy and mitotic arrests. The binding of other arsenic protein targets may also cause altered DNA repair enzyme activity, altered DNA methylation patterns and cell proliferation. (10, 3) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolism | Arsenic is mainly absorbed by inhalation or ingestion, and to a lesser extent by dermal exposure. It is then distributed throughout the body, where it is reduced into arsenite, then methylated into monomethylarsenic (MMA) and dimethylarsenic acid (DMA) by arsenite methyltransferase. Arsenic and its metabolites are primarily excreted in the urine. Arsenic is known to induce the metal-binding protein metallothionein, which decreases the toxic effects of arsenic and other metals by binding them and making them biologically inactive, as well as acting as an antioxidant. (5) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Values | LD50: 48 mg/kg (Oral, Rat) (11) LD50: 8 mg/kg (Intravenous, Rabbit) (12) LD50 in rabbits is 6 mg/kg (0.006 g/kg). | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lethal Dose | 1, carcinogenic to humans. (8) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Carcinogenicity (IARC Classification) | 1, carcinogenic to humans. (8) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Uses/Sources | Arsenic acid is used as a wood preservative, broad-spectrum biocide, and finishing agent for glass and metal. (9) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Minimum Risk Level | Acute Oral: 0.005 mg/kg/day (7) Chronic Oral: 0.0003 mg/kg/day (7) Chronic Inhalation: 0.01 mg/m3 (7) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Health Effects | Arsenic poisoning can lead to death from multi-system organ failure, probably from necrotic cell death and not apoptosis. Arsenic is also a known carcinogen, especially in skin, liver, bladder and lung cancers. (10, 5) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symptoms | Exposure to lower levels of arsenic can cause nausea and vomiting, decreased production of red and white blood cells, abnormal heart rhythm, and damage to blood vessels. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Treatment | Arsenic poisoning can be treated by chelation therapy, using chelating agents such as dimercaprol, EDTA or DMSA. Charcoal tablets may also be used for less severe cases. In addition, maintaining a diet high in sulfur helps eliminate arsenic from the body. (5) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Concentrations | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Links | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| DrugBank ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HMDB ID | HMDB0012190 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FooDB ID | FDB028842 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phenol Explorer ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| KNApSAcK ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BiGG ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BioCyc ID | ARSENATE | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| METLIN ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikipedia Link | Arsenate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemspider ID | 229 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ChEBI ID | 18231 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PubChem Compound ID | 234 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kegg Compound ID | C01478 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| YMDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ECMDB ID | ECMDB21394 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MSDS | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| General References | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||