Chlordecone (CHEM000128)
| Record Information | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Creation Date | 2009-03-06 18:58:09 UTC | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 2026-04-03 09:41:29 UTC | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Accession Number | CHEM000128 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Common Name | Chlordecone | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Small Molecule | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | Chlordecone is a manufactured insecticide also known as Kepone. It was used mainly on tobacco, ornamental shrubs, bananas, and citrus trees, and in ant and roach traps. However, its use was banned in 1975 due to its toxicity and nature as a persistent organic pollutant. (5, 6). The LC50 (LC = lethal concentration) is 0.022Р0.095 mg/kg for blue gill and trout. Kepone bioaccumulates in animals by a factors up to a million-fold. Workers with repeated exposure suffer severe convulsions resulting from degradation of the synaptic junctions. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Sources |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Type |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
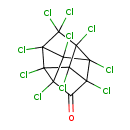
| Chemical Structure | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula | C10Cl10O | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Mass | 490.636 g/mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Mass | 485.683 g/mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS Registry Number | 143-50-0 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name | decachloropentacyclo[5.3.0.0²,⁶.0³,⁹.0⁴,⁸]decan-5-one | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional Name | kepone | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES | ClC12C(=O)C3(Cl)C4(Cl)C1(Cl)C1(Cl)C2(Cl)C3(Cl)C4(Cl)C1(Cl)Cl | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Identifier | InChI=1S/C10Cl10O/c11-2-1(21)3(12)6(15)4(2,13)8(17)5(2,14)7(3,16)9(6,18)10(8,19)20 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key | LHHGDZSESBACKH-UHFFFAOYSA-N | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | belongs to the class of organic compounds known as monoterpenoids. Monoterpenoids are compounds containing a chain of two isoprene units. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Organic compounds | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Lipids and lipid-like molecules | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Prenol lipids | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Monoterpenoids | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Monoterpenoids | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Aliphatic homopolycyclic compounds | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Status | Detected and Not Quantified | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Origin | Exogenous | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biofluid Locations | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tissue Locations | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathways | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Applications | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Roles | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Roles | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| State | Solid | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | White powder. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Profile | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Route of Exposure | Oral(5) ; inhalation (5) ; dermal (5). | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism of Toxicity | It is believed that the α-noradrenergic and serotonergic transmitter systems in the central nervous system are the primary neurotransmitter systems affected by chlordecone's neurotoxicity. Chlordecone causes spontaneous neurotransmitter release and increases in free intracellular calcium in synaptosomes by increasing permeability of the plasma membrane, activating voltage-dependent calcium channels, inhibiting of brain mitochondrial calcium uptake, and decreasing the activity of calmodulin-stimulated enzymes and Na+/K+, Mg+, and Ca+ ATPases. This inhibition of membrane ATPases also impairs energy-dependent cellular processes. Chlordecone causes its reproductive effects by binding to the estrogen and androgen receptors. (5, 1, 2) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolism | Chlordecone is well absorbed orally and through inhalation, but may also be absorbed dermally to a lesser extent. It is widely distributed throughout the body and concentrates in the liver, where it is metabolized to chlordecone alcohol by chlordecone reductase. Chlordecone, chlordecone alcohol, and their glucuronide conjugates are slowly excreted in the bile and eliminated in the feces. (5) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Values | LD50: 132 mg/kg (Oral, Rat) (9) LD50: 410 mg/kg (Percutaneous, Rabbit) (9) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lethal Dose | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Carcinogenicity (IARC Classification) | 2B, possibly carcinogenic to humans. (4) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Uses/Sources | Chlordecone is an insecticide. (5) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Minimum Risk Level | Acute Oral: 0.01 mg/kg/day (3) Intermediate Oral: 0.0005 mg/kg/day (3) Chronic Oral: 0.0005 mg/kg/day (3) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Health Effects | Chlordecone damages the nervous system, skin, liver, and male reproductive system. Animal studies indicate that it may also have harmful kidney effects, developmental effects, and effects on the ability of females to reproduce. (5) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symptoms | Exposure to chlordecone may cause tremors, jerky eye movements, memory loss, headaches, slurred speech, unsteadiness, lack of coordination, loss of weight, rash, enlarged liver, decreased libido, sterility, chest pain, arthralgia, and an increased risk of developing cancer. (6) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Treatment | Treatment of chlordecone exposure is symptomatic. (7) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Concentrations | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Links | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| DrugBank ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HMDB ID | HMDB0059603 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FooDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phenol Explorer ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| KNApSAcK ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BiGG ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BioCyc ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| METLIN ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikipedia Link | Kepone | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemspider ID | 293 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ChEBI ID | 16548 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PubChem Compound ID | 299 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kegg Compound ID | C01792 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| YMDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ECMDB ID | M2MDB005652 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MSDS | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| General References | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||